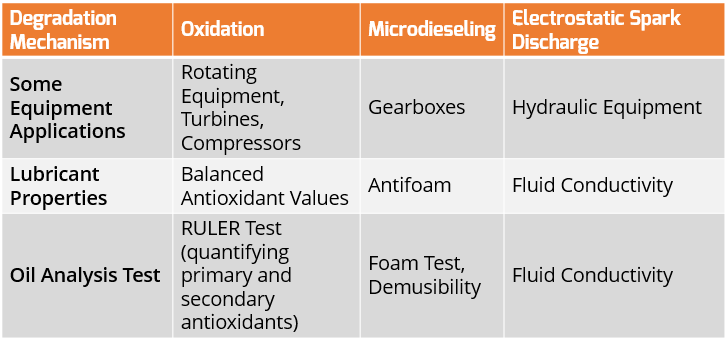
What Evidence is needed to prove that oxidation has occurred / is occurring?
However, understanding the oxidation process is just one part of the puzzle. When performing an investigation, we also need to know what factors or characteristics should be present. Additionally, we need to prove that their presence confirms our hypothesis of whether or not oxidation is occurring. This is where the line of evidence-based questioning plays a significant role.
When oxidation occurs, it is usual to see the presence of aldehydes, ketones, hydroperoxides, and even carboxylic acids. These can be confirmed using the FTIR (Fourier Transform Infrared test).
Typically, one will also find some deposits in the system. These deposits can be further characterized and tested to determine their nature using FTIR. Their presence, however, may be confirmed using the MPC (Membrane Path Colorimetry, ASTMD7843) test.
Identifying the presence of the deposits and/or the compounds listed above can lead to the conclusion that oxidation has occurred.
Another critical characteristic of oxidation is the depletion of antioxidants. This can be easily identified by utilizing the RULER® (Remaining Useful Life Evaluation Routine) test. This test quantifies the remaining antioxidants in the oil and gives the value for the amines and phenols (which is very important, especially in synergistic mixtures).
As such, one can detect the trend in the depletion of antioxidants and implement measures to prevent this before they become depleted.
The main tests to assist in determining the presence of Oxidation include:
RULER (Remaining Useful Life Evaluation Routine) levels less than 25% compared to new oil. This value represents the level of antioxidants in the oil. Hence, low levels indicate that the antioxidants are decreasing, possibly due to oxidation. This test can accurately give information on whether oxidation is currently occurring in the oil before deposits are formed.
An increase in acid number indicates the presence of acids resulting from oxidation. However, it must be noted that this change in acid number only occurs after oxidation has taken place. Hence this test is not a good indicator to determine if oxidation is occurring; instead, it is more definitive in letting us know that oxidation has already occurred.
Rapid color changes – darkening of the oil due to the deposits being present. While color is not the best indicator, in some instances, the darkening of the oil can provide a bystander to ask whether something is occurring in the oil. It is not a definitive test for the presence of oxidation.
FTIR test (Fourier Transform Infrared) for the presence of insolubles formed during the oxidation reaction. This can accurately determine the presence of any compound to assist us in determining whether oxidation is occurring.
MPC (Membrane Patch Colorimetry) levels outside the normal range (above 20). This lets us know that insoluble deposits are present in the oil. One must note that there may be instances where the deposits might not appear in the MPC test. As such, this should not be a standalone test to determine the presence of deposits.
RPVOT (Rotating Pressure Vessel Oxidation Test) levels are less than 25% compared to new oil (this is the warning limit). This is the industry standard, but this test does not have a high repeatability value in that if the same test were performed on identical samples, the values would be different. Additionally, the value (reported in minutes) is not easily translated into the environment of the components.
These tests provide us with the evidence we need to determine the presence of oxidation when performing the root cause analysis on the component’s failure.
Find out more in the full article, "Here's a more comprehensive approach to revealing oxidation root causes" featured in Precision Lubrication Magazine by Sanya Mathura, CEO & Founder of Strategic Reliability Solutions Ltd.